What is a vaccine?
According to the World Health Organization (WHO) “a vaccine is understood to be any preparation intended to generate immunity against a disease by stimulating the production of antibodies. This may be, for example, a suspension of killed or attenuated microorganisms, or products or derivatives of microorganisms. Most vaccines are given by an injection, but some are given orally (by mouth) or sprayed into the nose.”
In a previous article we had talked about the immune system, which is like the body’s defense army. One is born with a capacity to respond to what the body recognizes as foreign, as a threat (such as a virus, a bacterium, a fungus, or a parasite). That primary responsiveness is quick, but it is not specific and, therefore, sometimes not enough, the threat manages to get past that barrier. We call that first, fast and nonspecific immunity innate immunity.
The body has another way of defending itself, specially designed for each type of threat. Let’s say that the body has, from birth, a group of cells ready to design and produce specific weapons for each pathogen. This second army works on what we call acquired immunity.
Since there is a wide variety of pathogens, [1] these cells do not produce these specific weapons until the pathogen enters the body, they recognize it, and they can design exactly the weapon that will harm it. This response is more specific, but it takes longer to start working.
One of the most powerful weapons in this army are the antibodies (also known as immunoglobulins). Antibodies are molecules (glycoproteins) that synthesize cells of the immune system (lymphocytes), these antibodies are synthesized with the exact composition that allows it to specifically (very specifically) bind to a part of the pathogen.
Antibodies have two main functions: they mark these pathogens to be attacked and eliminated by other cells of the immune system, or they bind to a specific part of the pathogen that blocks its ability to enter and harm cells in the body.
Antibodies are generated against specific substances of the pathogens; these substances are called antigens. They are that part of the pathogen that, by interacting with the cells of the immune system, provokes the immune response, which is why identifying them is an important part in the production of vaccines.
Once this army that participates in acquired immunity, designs this production line against a specific pathogen, it already leaves it there programmed (immunological memory), so if this pathogen attacks us again all these specific weapons are ready, the response is faster, if you get sick it is usually much less serious, and many times you do not get sick because the immune system of your body fought the threat and eliminated it before it could harm you and cause symptoms.
That’s what a vaccine does, getting in contact with the pathogen (or parts of it), in a safe (non-disease-causing) way, but enough to trigger your immune response and leave all your weapons ready so if that pathogen attacks you naturally, your response is quick, specific and protective.
What types of vaccine are there?
There are different types of vaccines. Some of them contain the complete infectious agent (live attenuated vaccines and inactivated or dead vaccines). In live attenuated vaccines, an attenuated or weakened form of the disease-causing pathogen (such as chickenpox or smallpox vaccines) is used. So that it elicits a very strong immune response, most of these vaccines need a single dose to immunize you for life. However, when using an attenuated form of the pathogen it should be used with caution in people with weakened immune systems, and it has specificities for its preservation (they should always be kept cold).
Inactivated vaccines use the inactivated version of the pathogen (for example, against polio or rabies). They use a harmless version of the pathogen, but usually do not provide an immunity as strong as live vaccines, which is why multiple doses are often required.
On the other hand, vaccines with toxoids (against diphtheria and tetanus, for example) are those that use the toxins (toxic substances) released by the pathogen when they are the cause of the disease. It generates immunity against this harmful toxin, not against the pathogen itself.
There is another group of vaccines with greater biotechnological complexity: conjugate and recombinant vaccines. These employ fragments of the pathogen’s molecular structure, which elicit a protective immune response, which is the goal of all vaccines.
They are very safe vaccines, that can be used in anyone and offer a very strong immune response directed at key parts of the pathogen. Conjugates combine these parts of the infectious agent (virus or bacteria) with other molecules that increase their immunogenic capacity (for example, vaccines against some meningococci and pneumococci), while recombinants (such as vaccines against hepatitis B, human papilloma virus or herpes zoster) involves introducing into any vector―it is usually a virus or bacterium that does not cause disease―regions of the pathogen that we know to be immunogenic; that is, they have the capacity to activate the immune system.
Among the novel techniques being used for the production of vaccines are DNA vaccines, nanoparticle vaccines, among others.
Those involving genetic engineering, the so-called DNA vaccines, have had a major boost with technological development that has succeeded in sequencing (knowing the genetic information) of many pathogens very quickly. The sequence of the current coronavirus, for example, was obtained in just days. Researchers use an organism’s genome (its genetic information) to extract the genes that are most likely to match known antigens that could be used in a vaccine.
Once identified, those genes can be combined and inserted into a different, rapidly multiplying organism, such as yeast, to produce experimental antigens, which are then studied to determine their ability to elicit a protective immune response. This method is known as “reverse vaccination”; no licensed vaccine has yet been released, but several experimental vaccines are already being studied, some of which are in the later stages of clinical trial (for example, a group B meningococcal vaccine). [2] Several of the vaccine candidates against COVID-19 follow this method.
What process does a vaccine candidate have to follow until it is approved for use in humans?
The creation of a vaccine is a long and complex process that often takes 10 to 15 years, and involves the combined participation of governments, and public and private organizations.
The World Health Organization establishes a protocol that many governments and regulatory institutions in the world follow, although each of them has specific regulations.
Ensuring that vaccines are safe, effective and of quality is a crucial element in their development and distribution. It begins with the first phases of the vaccine, generally in the laboratory, where its components are subjected to tests to determine aspects such as purity and potency. The clinical trials consisting of three phases are then commenced.
The license, or authorization for use in humans is the fundamental step in the process. The official entity that grants the authorization, the national regulatory body is the arbitrator that decides whether the established standards have been met to guarantee the quality of the vaccine.
What are the steps that have to be followed?
Exploration stage
This stage involves basic laboratory research, and often lasts 2 to 4 years.
Preclinical stage
Preclinical studies use tissue culture or cell culture systems and animal testing to assess the safety of the candidate vaccine and its ability to elicit an immune response.
Researchers can tailor the candidate vaccine during the preclinical phase to try to make it more effective. They can also perform exposure studies on animals, which means animals are vaccinated and then they try to infect them with the target pathogen; these types of studies are never performed on humans.
Many candidate vaccines do not go beyond this stage, as they cannot elicit the desired immune response. Often the preclinical stages last 1 to 2 years.
To continue the studies, after completing this phase, an application must have been approved by a competent agency.
Human clinical studies
Phase I
This first attempt to evaluate the candidate vaccine in humans involves a small group of adults, generally between 20 to 80. If the vaccine is aimed at children, the researchers will first test it in adults, and will gradually reduce the age of the test subjects until they reach the target. The goals of phase I trials are to assess the safety of the candidate vaccine and to determine the type and extent of the immune response that the vaccine elicits.
Phase II
A larger group of several hundred people participates in phase II testing. Some of the people may belong to groups at risk of contracting the disease; the trials are randomized and well controlled, and include a placebo group. The goals of phase II trials are to study the candidate vaccine for its safety, immunogenicity, proposed doses, vaccination schedule, and method of application.
Phase III
Candidate vaccines that are successful in phase II advance to larger trials, involving thousands to tens of thousands of persons. Phase III trials are randomized and double-blind, and involve the experimental vaccine that is tested against a placebo (the placebo may be a saline solution, a vaccine for another disease, or some other substance). One of phase III’s goals is to evaluate the safety of the vaccine in a large group of persons. Some unusual side effects may not be apparent in smaller groups of people who were part of the previous phases.
During these phases, the efficacy of the vaccine to protect against the disease is assessed. Tests are done that have to do with the production of antibodies and the immune response of the persons who receive the vaccine. After a phase III trial is successful, accredited agencies will inspect the product, the factories and research results, until approval is issued.
After approved for large-scale use, the vaccines continue to be monitored.
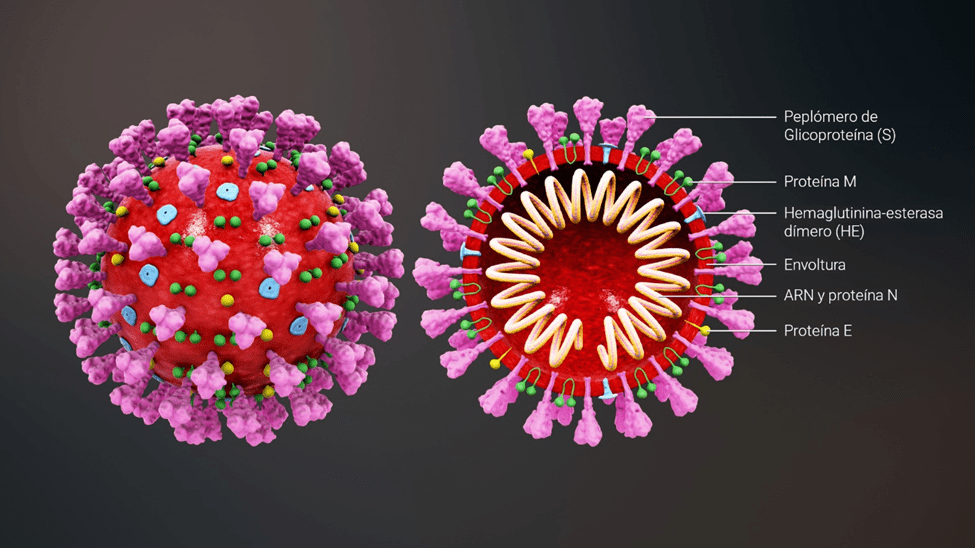
Structure of the SARS-Cov-2 coronavirus
SARS-COV-2 is an enveloped, RNA-positive virus. The “key” to enter the cell is found in the so-called “spike” proteins (S), which cover the virus envelope.
SARS-CoV-2 coronavirus vaccines and treatments
The process to start a vaccine can take many years, however, we are told that probably in just over a year we can have a vaccine against this new virus. A response that, if possible, would be of a speed never seen before against a new disease.
This is mainly due to advances in the biotechnology sector. First of all, just one week after China reported the first cases of severe pneumonia of unknown origin to the WHO, the causative agent―the new SARS-CoV-2 coronavirus―was identified. A few days later its genome was already available. In just under three months, more than 970 scientific articles are available in the PubMed database.
Knowing the biology of the virus facilitates the design of therapeutic (antiviral) and preventive (vaccines) strategies. The similarity of genetic information with another coronavirus that has been studied for years, SARS-Cov, which caused the epidemic of acute respiratory syndrome (SARS) in 2002, has led to rapid progress in the pre-clinical phases.
In just these three months there are already several therapeutic proposals and vaccine candidates against the new coronavirus. Science has never advanced so far in such a short time to combat an epidemic. Many of the proposals come from research groups that have spent years working against other viruses, especially against SARS and MERS. This accumulated knowledge has now made it possible to go at a speed never seen before.
Antiviral therapies
Some already available antiviral drugs have been tested to see if they can be effective in fighting COVID-19. Chloroquine, which has been used for years against malaria, is being studied by a group of researchers, as it could reduce the viral load by blocking the virus from entering cells. Some anti-inflammatories, such as barcitinib and mesmosate from camostat (Japan), are being used in some protocols because they could block the entry of the virus into lung cells.
One of the most promising antivirals against SARS-CoV-2 is remdesivir, an inhibitor that prevents the virus from multiplying within the cell. It has already been used against SARS and MERS and has been successfully tested in the latest Ebola epidemics, and against other viruses with the RNA genome. It is, therefore, a broad-spectrum antiviral. At least twelve phase II clinical trials are already underway in China and the U.S., and another has started in phase III with 1,000 patients in Asia.
In the United States, in New York, the FDA has approved the use of plasma from sick patients who have recovered. This involves obtaining blood from donors who have recovered from COVID-19, and isolating the plasma (where the antibodies are located), to transfuse it to sick people. It is not a new treatment; it was used in the Spanish Flu pandemic in 1918. According to the journal Nature, this effort in the United States is following preliminary studies carried out in China. The convalescent plasma approach has also had modest success during previous outbreaks of severe acute respiratory syndrome (SARS) and Ebola. It could be an emergency response in which more effective treatments appear.
There are at least 27 clinical trials with different combinations of antiviral treatments such as Interferon Alfa-2B, ribavirin, methylprednisolone, and azvudine. At the moment they are experimental treatments, but they are a hope for the most serious and severe cases.
COVID-19 vaccines for the future
The main hope for controlling the disease is based on achieving effective vaccines. The WHO, until March 20, had a list of 41 candidates, but based on press reports from various countries, we know that more are being worked on.
An article published on March 23 by The Conversation summarizes some of the most promising projects.
In clinical trial phase
According to the publication, one of the most advanced is the Chinese proposal, a recombinant adenovirus vector-based vaccine with the SARS-CoV-2 S gene, which has already been tested in monkeys and is known to produce immunity. A phase I clinical trial will be started with 108 healthy volunteers, between 18 and 60 years old, in which three different doses will be tested.
Other proposals are being promoted by CEPI (Coalition for Epidemic Preparedness Innovations), an international association in which public, private, civil and philanthropic organizations collaborate to develop vaccines against epidemics. It is currently funding eight SARS-CoV-2 vaccine projects that include recombinant, protein, and nucleic acid vaccines.
mRNA-1273 vaccine (Moderna, Seattle)
It is a vaccine made up of a small fragment of messenger RNA with the instructions to synthesize part of the protein S of the SARS-Co-V. The idea is that, once introduced into our cells, it is these cells that make this protein, which would act as an antigen and stimulate the production of antibodies. It is already in the clinical phase and it has begun to be tested in healthy volunteers.
Preclinical phases
Recombinant measles virus vaccine (Pasteur Institute, Themis Bioscience and University of Pittsburg)
It is a vaccine built on a live attenuated measles virus, which is used as a vehicle and contains a gene that encodes a protein of the SARS-CoV-2 virus. It is in the preclinical phase.
Recombinant Influenza Virus Vaccine (University of Hong Kong)
It is also a live vaccine that uses an attenuated influenza virus as a vector, which has had the virulence gene NS1 removed, and is therefore not virulent. A SARS-CoV-2 virus gene is added to this vector virus. This proposal has some advantages: it could be combined with any strain of seasonal influenza virus and thus serve as a flu vaccine, it can be quickly manufactured in the same production systems that already exist for influenza vaccines, and they could be used as intranasal vaccines via spray. It is in the preclinical phase.
Recombinant protein vaccine obtained by nanoparticle technology (Novavax)
This company already has vaccines against other respiratory infections such as adult flu (Nano-Flu) and respiratory syncytial virus (RSV-F) in clinical phase III and has manufactured vaccines against SARS and MERS. Its technology is based on producing recombinant proteins that are assembled into nanoparticles and administered with its own adjuvant, Matrix-M. This compound is a well-tolerated immunogen capable of stimulating a powerful and long-lasting nonspecific immune response. The advantage is that in this way the number of necessary doses would be reduced (thus avoiding revaccination). It is in the preclinical phase.
Recombinant vaccine using as a vector the Oxford chimpanzee adenovirus, ChAdOx1 (Jenner Institute, Oxford University)
This attenuated vector is capable of carrying another gene that encodes a viral antigen. Models for MERS, influenza, chikungunya and other pathogens such as malaria and tuberculosis have been tested in volunteers. This vaccine can be manufactured on a large scale in bird embryo cell lines. The recombinant adenovirus carries the glycoprotein S gene of the SARS-CoV-2. It is in the preclinical phase.
Recombinant Protein Vaccine (University of Queensland)
It consists of creating chimeric molecules capable of maintaining the original three-dimensional structure of the viral antigen. It uses the technique called “molecular clamp,” which allows vaccines to be produced using the virus genome in record time. It is in the preclinical phase.
Messenger RNA Vaccine (CureVac)
This is a proposal similar to that developed by the modern biotechnology company, with recombinant messenger RNA molecules that are easily recognized by the cellular machinery and produce large amounts of antigen. They are packaged in lipid nanoparticles or other vectors. In preclinical phase.
DNA INO-4800 vaccine (Inovio Pharmaceuticals)
It is a platform that manufactures synthetic vaccines with DNA of the S gene from the surface of the virus. They had already developed a prototype against MERS (the INO-4700 vaccine) that is in phase II. They recently published the phase I results with this INO-4700 vaccine and found that it was well-tolerated and produced a good immune response (high antibody levels and a good T-cell response, maintained for at least 60 weeks after vaccination). In preclinical phase.
Cuba
According to the director of biomedical research of the CIGB, Gerardo Guillén, the Center for Genetic Engineering and Biotechnology (CIGB) of Cuba has a vaccine design that could be used against the new coronavirus.
According to the Cuban scientist, this vaccine is in the methodological and design phase. However, according to his statements, there is an advanced path since a platform that the institution has already developed is being used, where it works with virus-like particles with great capacity to stimulate the immune system.
Another platform that is very attractive and promising being developed by the center is by immunization through the nose. Cuba has experience in this regard, since it has a registered vaccine that uses this nasal spray.
The Cuban vaccine candidate is being developed with the Cuba-China joint research and development center, located in Hunan province. It is not known when clinical trials could begin.
Cuba is also carrying out research in therapeutic drugs. The results so far published by China in the treatment of COVID 19, with the Cuban Interferon Alfa 2B, showed positive results.
***
All proposals for specific treatments and vaccines for COVID-19 are in the experimental phase. But technological advances and the accumulation of research results in the fields of antiviral therapies and vaccines against other viruses, and specifically against other coronaviruses, make many experts affirm that there is a high probability of success. Although we want and need faster responses, science cannot be asked to have a vaccine in less than a year, in reality that would already be a record time.
The international scientific community’s actions, in terms of sharing scientific results, collaboration and training, is the backbone of this battle, and my greatest hope.