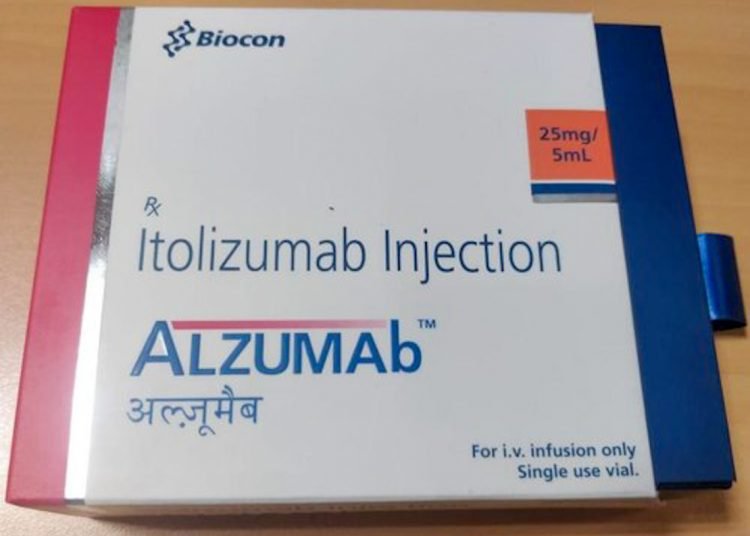
The Cuban drug Itolizumab, regularly used for the treatment of psoriasis and which has been shown to be effective within the Cuban protocol against COVID-19, received the approval of the Indian Regulatory Office of Medicines for the treatment in case of emergency of coronavirus patients with moderate to severe acute respiratory distress.
The General Controller of Medicines, Dr. VG Somani, stated that the use of the medicine will be limited to Indian patients with severe symptoms, since said treatment is applied when cytokine release syndrome occurs, causing patients to present acute or severe respiratory distress.
The approval of the Cuban monoclonal drug has been included since July 10 in India’s protocol, according to the PTI press agency, quoted in a note by the island’s Agencia Cubana de Noticias news agency.
Despite being used for the first time against COVID-19, Itolizumab was in regular use in India for the treatment of psoriasis. For the use of the drug, each coronavirus patient must previously submit a written informed consent, said Dr. Somani.
India is currently one of the nations most affected by the SARS-CoV-2 pandemic, with an increase of tens of thousands of new cases in recent days, according to press reports.
Itolizumab is a product of the Cuban Center for Molecular Engineering. The Cuban specialists worked together with researchers from the company Biocon Asia, which obtained the license for its use as an active drug from the start.
A total of six hospitals in the Asian nation conducted clinical trials of the Cuban drug, to then incorporate it into their health protocols and deal with COVID-19, approval given by a committee made up of pulmonologists, pharmacologists and medical specialists from the Indian Institute of Medical Sciences.
La India, decidida a lanzar su vacuna contra la COVID-19 a mediados de agosto
Itolizumab received one of the National Awards of the Cuban Academy of Sciences 2014, in the Biomedical Sciences section, and since then it has also been used in various clinical trials.
There are several Cuban medicines being successfully used in the fight against the new coronavirus, among which are Interferon alfa 2B, used in China and other nations with good results, and more recently CIGB-258 or Jusvinza, used as Itolizumab in treating critically and seriously ill patients.