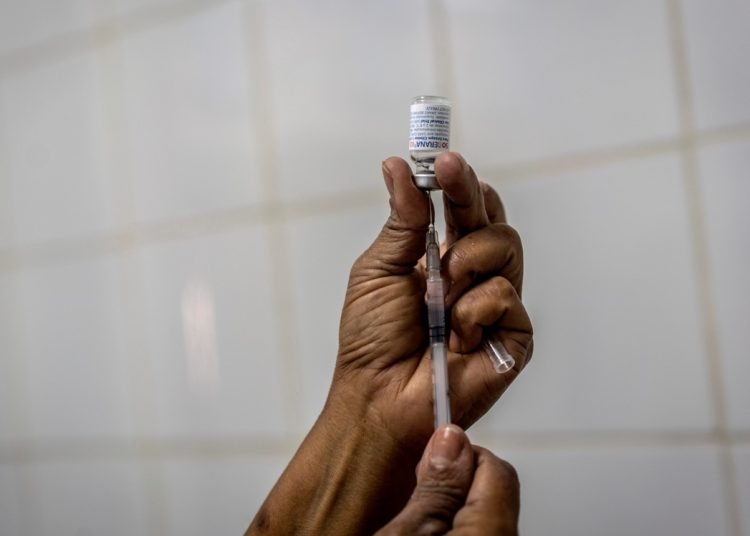
The Cuban biopharmaceutical industry already produces single-dose batches of its COVID-19 vaccine candidates without thiomersal, for the benefit of people allergic to this substance.
In this way, both Soberana 02 and Abdala, the most advanced COVID-19 vaccines on the island currently in phase III of clinical trials, can be inoculated to allergy sufferers, who until now have not been able to participate in the studies due to the use of thiomersal or thimerosal as a preservative in multi-dose presentations.
“People allergic to thiomersal can opt for getting vaccinated. There will be a vaccine for everyone,” Dr. Dagmar García Rivera, research director of the Finlay Vaccine Institute (IFV), said to the official Cubadebate website.
The specialist explained that the pharmaceutical industry frequently uses multi-dose batches of vaccines in which thiomersal is used as a preservative, “to guarantee the preservation of the sterility of the bulb that, necessarily, will be used several times to administer each of the doses.”
This has happened with the Cuban COVID-19 vaccine candidates, in particular with the most advanced ones, but now work is underway in the production of single-dose batches without this substance.
“In the case of Soberana 02, the multi-dose presentations always contain thiomersal. The first single-dose batches that were produced also contain thiomersal, precisely because they are formulations still in the stage of pharmaceutical development and small-scale productions,” said García Rivera.
However, she explained that as part of the productive scaling taking place in the National Center for Biopreparations (BioCen), “the last single-dose batches that have been produced and all those that will be produced from now on, will not contain thiomersal” and confirmed that in the intervention study that is being carried out in Havana with Soberana 02—a vaccine developed precisely by the IFV—and in which health and science workers are being immunized, among other sectors, batches have already been used without that substance.
More than 100,000 Cuban volunteers receive first dose in studies on COVID-19 vaccines
For her part, Dr. Marta Ayala Ávila, general director of the Center for Genetic Engineering and Biotechnology (CIGB), also confirmed to Cubadebate that work is already underway at the production scale level in doses without thiomersal of the Abdala vaccine candidate, “for future stages of clinical development.”
“At the moment the doses produced with thimerosal or thiomersal are being used, and doses are being prepared without this compound,” said the specialist, who also explained that in the case of the Mambisa vaccine candidate, which is inoculated through the nasal route and which concluded the phase I trials, this does not contain the substance that causes allergies in some people.
Cuba has five COVID-19 vaccine candidates understudy in humans and, so far, Cuban specialists evaluate the development of the tests as positive. In the case of clinical trials, these seek to measure the safety, efficacy and immunogenicity of the formulations, while controlled interventions are intended to evaluate the direct and indirect effects of vaccination against SARS-CoV-2, in population cohorts at risk of infection, disease and spread of the epidemic.
Cuban authorities recently announced a roadmap that includes the application of the two most advanced potential vaccines—Soberana 02 and Abdala—to hundreds of thousands of people in the next three months, under the figure of population intervention studies. Among them is one in Havana that plans to administer one of the two vaccine candidates to 1.7 of the 2.2 million inhabitants of the capital, the current epicenter of the pandemic on the island.