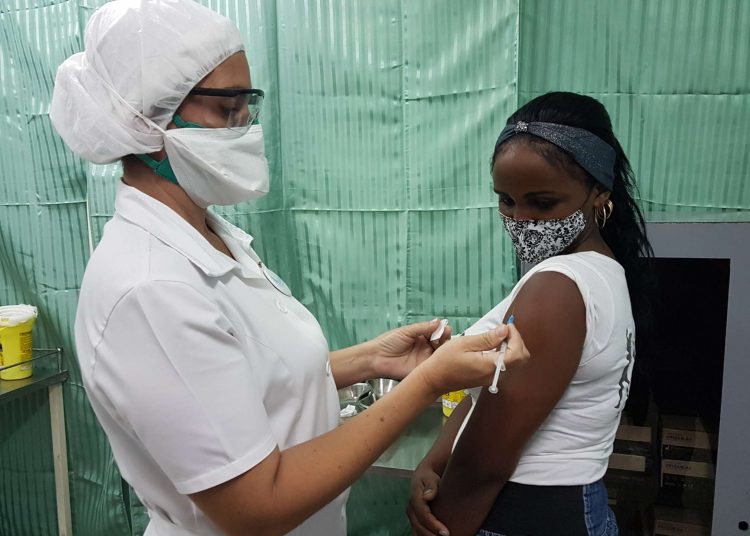
After concluding the application of its three doses in the more than 48,000 participants in phase III clinical trial in eastern Cuba, the evaluation of the efficacy of the Abdala COVID-19 vaccine candidate began this Monday.
The evaluation is headed by health professionals and the Center for Genetic Engineering and Biotechnology (CIGB), an institution in charge of the development and study of the immunogen, according to a report by the Agencia Cubana de Noticias (ACN)news agency.
Francisco Hernández, head of the clinical study, explained to the agency that at this stage “there will be a follow-up of all volunteers in order to detect positive symptomatic cases infected with the SARS-CoV-2 virus, which causes the systemic disease.”
A total of 48,290 people from the cities of Bayamo, Santiago de Cuba and Guantánamo participated in the study, of which more than 97% received the three doses of the drug, “a figure that shows adherence to the immunization protocol,” says the source.
Cuba: Inicia análisis de eficacia de candidato vacunal Abdala https://t.co/5tiCi9SKD9 a través de @YouTube
— BioCubaFarma (@BioCubaFarma) May 3, 2021
The follow-up of the volunteers “will make it possible to determine the proportions of the research subjects who fell ill according to the group: placebo or vaccinated,” and “constitutes an effective way to measure the main variable of the phase,” said Hernández, who detailed that “the efficacy results will depend on the delay in the appearance of volunteers infected with the coronavirus as estimated in the research design, a total of 150, although intermediate cuts are foreseen for every 50 infected of them.”
The specialist explained that the Joaquín Castillo Duany Military Hospital, in Santiago de Cuba, will take care of all the patients with COVID-19 who participated in the clinical trial in the three eastern provinces, where the three doses of the Abdala vaccine candidate were applied, while the Veguitas isolation centers, in Granma, and the Mariana Grajales Vocational School, in Guantánamo, will receive positive cases, contacts and suspects.
Hernández asked the participants in phase III of the studies “to maintain individual care because some received the vaccine and others the placebo, hence all are not protected,” says the ACN, according to which the expert also insisted on the importance of reinforcing sanitary and protection measures in the current epidemiological scenario and stressed that, although vaccines do not prevent coronavirus infection, “one of their purposes is to prevent the development of serious and severe forms of the disease and, consequently, deaths from COVID-19.”
Abdala is now also being applied to health personnel and other risk groups in intervention studies carried out in Havana and in eastern Cuba. In addition, it will be part, together with Soberana 02, of the massive intervention that should begin in May in the capital and in which the authorities intend to immunize 1.7 million people.
Soberana 02, the other most advanced Cuban COVID-19 vaccine candidate, led by the Finlay Vaccine Institute (IFV), is currently in phase III of clinical trials in the capital, in which 44,010 volunteers aged from 19 to 80 are participating, and it will also be tested in Iran, where 100,000 doses of the formula have been sent.
In addition, the island has three other candidates in the human testing phase: Mambisa, also from the CIGB and which is applied intranasally, as well as Soberana 01 and Soberana Plus, both from the IFV. The latter, intended for people who have already suffered from COVID-19, is in phase II of clinical trials with 450 convalescents.